 
On the other hand, the copper atom donates an electron in the 4s orbital and an. 28 Nickel, Ni, 1s22s22p63s23p63d84s Ar3d 4s 4p 29 Copper, Cu, 1s22s22p63s23p63d104s Ar3d 4s 4p 30 Zinc, Zn, 1s22s22p63s23p63d104s Ar3d 4s 4p 31. Therefore, one of the 4s 2 electrons of the Copper (Cu) atom jumps to the 3d 9 to achieve a fully -filled 3d orbital. Here, the electron configuration of copper ion(Cu+) is 1s2 2s2 2p6 3s2 3p6 3d10. However, half– filled or fully-filled orbitals are more stable due to the symmetrical distribution of electrons. The expected electronic configuration of Copper (Cu): 1s 22s 22p 63s 23p 6 4s 23d 9 Thus, the expected electronic configuration of Copper (Cu) will be as follows: By following the same rule of the Aufbau Principle, within the remaining 11 electrons, two electrons are placed in the 4s orbital, and 9 electrons are placed in the 3d orbital.Further, the next 6 electrons will go to the 3p orbital.Then, the next two electrons go in the 3s.  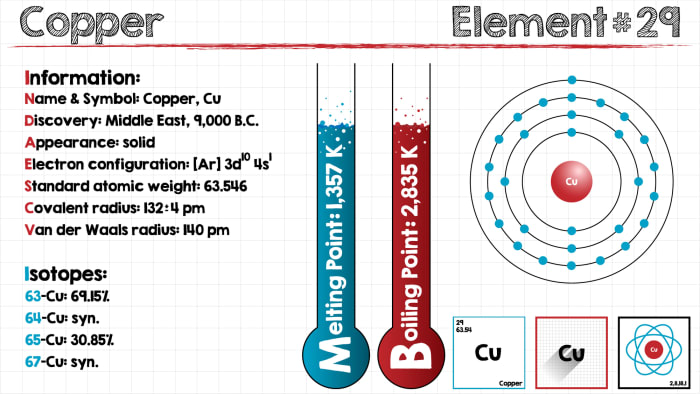
And, the next 6 electrons for Copper (Cu) will go to the 2p orbital.Then, the next two electrons for Copper (Cu) occupy the 2s orbital.According to Aufbau Principle, the first two electrons of Copper (Cu) are filled in 1s orbital.According to Aufbau Principle, “the orbitals are filled up with electrons in order of their increasing energy, i.e., orbital of lowest energy are filled up first, and those with the highest energy are filled up at the end.” The order of the orbital occupancy is shown in the following diagram. So, let’s start writing the electronic configuration of Copper (Cu) based on the Aufbau Principle. Electronic Configuration of Copper Electronic Configuration of Copper Atom (Cu) (Atomic Number 29) This article explains the Electronic Configuration of Copper and how to write the Electronic Configuration of Copper atom (Cu). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
